Kwangdong Pharmaceutical surged to the daily upper limit following news that a presbyopia treatment for which it holds exclusive domestic distribution rights, received marketing approval from the U.S. Food and Drug Administration (FDA).
U2Bio also hit the daily price limit, driven by expectations of enhanced corporate value stemming from the spin off of its clinical trial services and biotechnology divisions as well as a strategic partnership with Daewoong.
CorestemChemon saw its stock rise by double digits amid speculation that its domestic marketing approval for a treatment for amyotrophic lateral sclerosis (ALS) could be maintained.
◇Kwangdong Pharmaceutical Secures FDA Approval for Presbyopia Treatment with Exclusive Domestic Rights
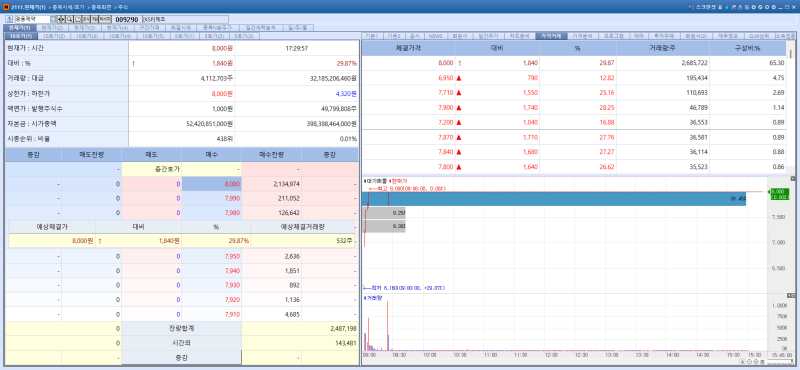
According to KG Zeroin’s MP DOCTOR (formerly MarketPoint), Kwangdong Pharmaceutical’s shares surged 29.87% from the previous trading day to close at 8000 won. The rally was attributed to news that YUVEZZI, a presbyopia treatment for which Kwangdong holds exclusive domestic distribution rights, received marketing approval from the U.S. Food and Drug Administration (FDA), raising expectations for its swift introduction to the Korean market.
In September last year, Kwangdong applied to South Korea’s Ministry of Food and Drug Safety (MFDS) for marketing approval of YUVEZZI, and the review process is currently underway. YUVEZZI is a novel drug developed by global biotech company Tenpoint Therapeutics and was previously known as BRIMOCHOL PF during development.
In January 2024, Kwangdong signed an exclusive licensing agreement with Hong Kong-based Zhaoke Ophthalmology, which holds Asian rights to BRIMOCHOL. YUVEZZI is a fixed-dose combination of carbachol (2.75%) and brimonidine tartrate (0.1%).
The drug is known as the first and only FDA-approved dual-action eye drop for presbyopia. It works by constricting the pupil to create a pinhole effect, thereby improving near vision and depth of focus. When administered once daily, its effects begin within 30 minutes and can last up to 10 hours.
The FDA approval was based on data from two Phase 3 clinical trials (BRIO I and BRIO II) involving more than 800 patients. The trials demonstrated statistically significant improvement in binocular uncorrected near visual acuity without compromising distance vision. YUVEZZI also established safety and tolerability over a 12-month study period, the longest conducted to date in the presbyopia eye drop category.
Kwangdong is fostering ophthalmology as a next-generation growth engine and expanding its portfolio in the field. In addition to YUVEZZI, the company has secured exclusive domestic rights to several innovative treatments including Raxone for rare ophthalmic diseases, OCU400 for retinitis pigmentosa and NVK002 for pediatric myopia.
A company official stated that YUVEZZI’s approval under the FDA’s stringent standards is an encouraging sign for its domestic introduction, adding that the company will make every effort to complete the remaining approval procedures and prepare for market launch to provide a new treatment option for Korean patients and physicians.
◇U2Bio Rises on Strategic Partnership with Daewoong and Corporate Spin Off
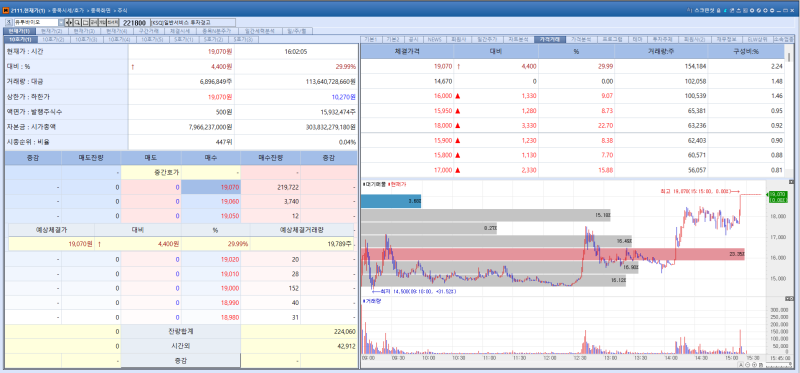
U2Bio’s shares jumped 29.99% to 19,070 won. The surge was driven by expectations of growth following a strategic partnership with Daewoong and a corporate spin-off.
Daewoong announced on the 12th that it would dispose of 564,745 shares of treasury stock through an in-kind contribution to U2Bio for strategic investment purposes. The transaction price is reportedly 21,500 won per share, with a total value of approximately 12.1 billion won.
Under the deal, U2Bio will issue 2,388,278 new shares through a third-party allotment to acquire Daewoong’s treasury shares. Upon completion of the share swap, U2Bio will hold a 0.97% stake in Daewoong, while Daewoong will own a 14.99% stake in U2Bio, becoming its second-largest shareholder. Through this collaboration, Daewoong plans to accelerate investments in the digital healthcare sector.
Separately, U2Bio is proceeding with a physical spin-off involving Jigoo Holdings. The surviving entity will be Jigoo Holdings, which will operate the contract research organization (CRO) and biotechnology businesses, while the newly established U2Bio will focus on its remaining operations, including in vitro diagnostics services and medical IT solutions.
As the spin-off will be conducted through a simple physical division in which 100% of the shares issued by the new entity are allocated to the surviving company, no split ratio will be calculated, and there will be no change in the major shareholder’s ownership before and after the division.
The company explained that the restructuring aims to concentrate U2Bio’s capabilities on its diagnostics and medical IT businesses to promote independent growth, while strengthening Jigoo Holdings’ healthcare operations. It added that the improved governance structure is expected to enhance overall growth, profitability, corporate value, and shareholder value.
◇CorestemChemon Gains on Possibility of Maintaining ALS Drug Approval
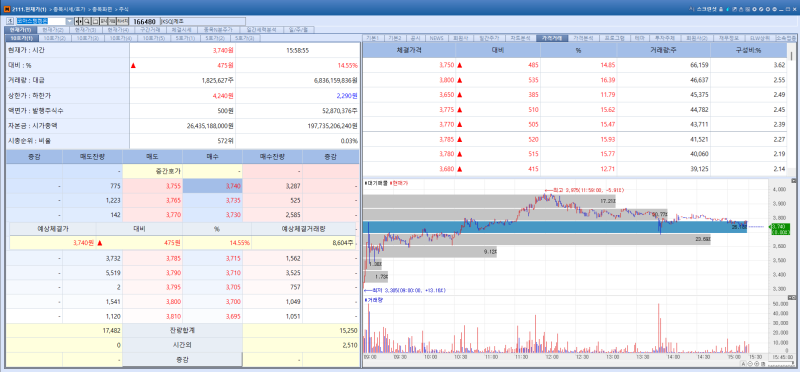
CorestemChemon’s shares rose 14.55% to 3,740 won. The increase followed speculation that the company may retain domestic marketing approval for its amyotrophic lateral sclerosis (ALS) treatment, Neuronata-R.
According to a premium pharmaceutical and biotech report published by Edaily, the MFDS is expected to soon announce its decision regarding a change in marketing authorization for Neuronata-R. The decision is also significant as it effectively serves as a precursor to a potential Biologics License Application (BLA) submission to the FDA in the United States.
Since applying for a modification of its marketing approval in April last year, CorestemChemon has submitted supplementary data multiple times in close consultation with the MFDS. The company confirmed that the regulator no longer requires additional data related to Neuronata-R, suggesting that major administrative hurdles have largely been resolved.
Neuronata-R originally received conditional approval from the MFDS in 2014 and has since been marketed to both domestic and international patients. However, the drug failed to meet its primary endpoint in a Phase 3 trial announced in December 2024. Despite this setback, the company maintains expectations for continued approval based on biomarker driven clinical significance.
Similar precedents exist. Mitsubishi Tanabe’s edaravone (Radicava) initially failed to meet the primary endpoint in the overall patient population but later secured approval based on subgroup analyses. Biogen’s tofersen (Qalsody) also did not achieve its primary endpoint yet obtained FDA accelerated approval based on improvements in the neurofilament light chain (NfL) biomarker.
The upcoming decision is expected to mark a critical turning point, affecting not only Neuronata-R’s continued domestic sales but also CorestemChemon’s global strategy.
A company representative stated that Neuronata-R has demonstrated the potential to extend survival and alleviate symptoms through its unique multi mechanism approach involving neuroprotection and inflammation control via stem cells.
The representative added that the therapy aligns with global precedents in ALS treatment and, given its distinct mechanism, may offer synergistic benefits when used in combination with standard treatments such as riluzole.










