Curacle’s stock rose after the company announced that it had signed a technology export deal worth up to 1.5 trillion won for a dual antibody drug candidate targeting retinal diseases which it is jointly developing with biotech venture Maptics.
Seegene’s share price also gained momentum as improved first quarter earnings driven by growth in its non-respiratory product lineup, boosted investor sentiment. Meanwhile Osang Healthcare’s stock climbed on expectations surrounding the development of its next-generation molecular diagnostics platform.
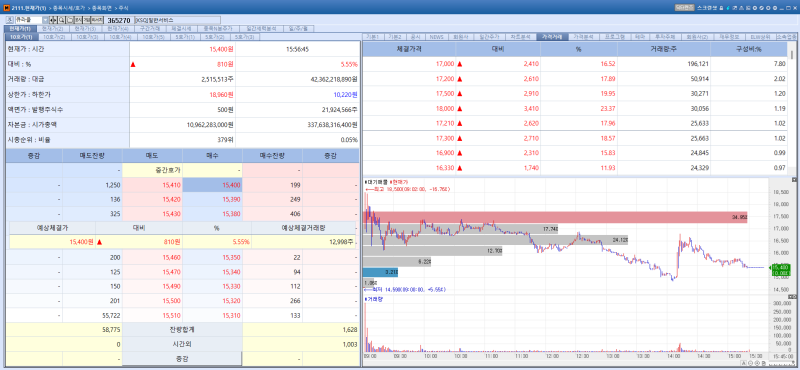
Curacle stock trend on May 11.(Image=MP Doctor)
◇Curacle Exports Retinal Disease Bispecific Antibody Drug Candidate to U.S. based NewCo Firm
According to KG Zeroin’s MP DOCTOR Curacle shares closed at 15,400 won on the day up 5.55% from the previous session. The gain marked the company’s second consecutive day of stock price increases. Curacle announced that it had licensed out MT-103 a bispecific antibody drug candidate for retinal diseases jointly developed with Maptics to U.S. based Memento Medicine.
The total deal size amounts to as much as $1.077 billion (approximately 1.6 trillion won) including an upfront payment of $8 million (about 11.8 billion won) and milestone payments. The transaction is regarded as surpassing the scale of Alteogen’s technology export deals signed in January and March this year. Under the agreement Curacle and Maptics will split the proceeds equally on a 50-50 basis.
Memento Medicine is a NewCo company established with participation from global venture capital firms and investors. A NewCo model involves creating a separate corporation centered on a specific asset while attracting outside capital to advance drug development.
Such companies are known for streamlined organizations led by specialized professionals, rapid decision making and focused investment in a limited number of pipelines to improve development efficiency. Memento Medicine will be responsible for the future development and commercialization of MT-103.
Curacle and Maptics, both focused on developing treatments for intractable vascular and metabolic diseases have been developing MT-103 to treat retinal vascular disorders such as wet age related macular degeneration and diabetic macular edema which occur when retinal blood vessels become unstable.
Excessive activity of vascular endothelial growth factor (VEGF) can lead to abnormal blood vessel formation and vascular leakage, eventually causing vision impairment.
MT-103 is a bispecific antibody candidate designed to inhibit VEGF while simultaneously activating Tie2, a receptor involved in vascular stabilization. While existing blockbuster drug Eylea focuses solely on anti-VEGF activity, MT-103 not only suppresses VEGF but also promotes Tie2 signaling to stabilize blood vessels.
In preclinical studies, MT-103 demonstrated superior efficacy in suppressing vascular leakage and abnormal blood vessel formation compared with Eylea and Vabysmo, another bispecific antibody treatment. The findings were presented in an oral session at ARVO 2026 the world’s largest ophthalmology conference held in Denver on May 3.
According to GlobalData the market for treatments targeting wet age related macular degeneration and diabetic macular edema is projected to grow from $19 billion (approximately 28 trillion won) in 2025 to $34 billion (about 50 trillion won) by 2031.
Yoo Jae hyun CEO of Curacle said “Global healthcare investors have joined the development of MT-103 and we expect the project’s development and commercialization to proceed quickly and systematically based on their financial resources and development expertise.”
He added “We believe this will become another successful example of the NewCo model which has recently attracted significant attention in the pharmaceutical and biotech industry.”
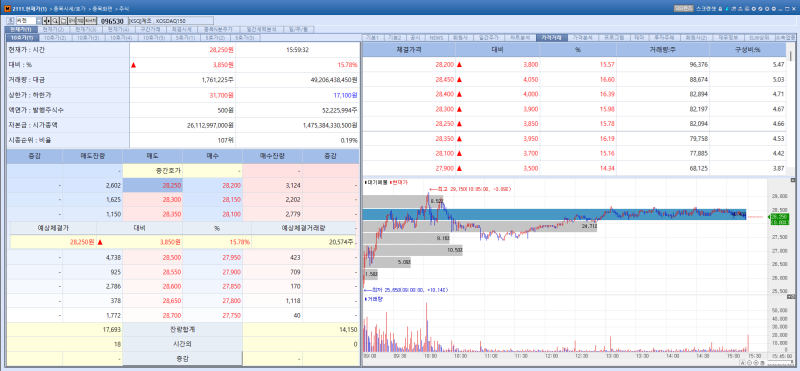
Seegene stock trend on May 11.(Image=MP Doctor)
◇Seegene Posts Improved First Quarter Earnings on Growth in Non respiratory Product Lineup
Seegene shares surged 15.78% from the previous trading session to close at 28,250 won on the day. The stock rally was attributed to improved first quarter earnings driven by strong growth in the company’s non respiratory product lineup.
Seegene reported first quarter revenue of 129.1 billion won, up 11.3% year on year. Operating profit jumped 58.6% from a year earlier to 23.6 billion won during the same period.
Growth in non respiratory syndromic diagnostic products led the earnings improvement. Revenue from syndromic diagnostic products increased 32.6% year on year. Syndromic diagnostics refer to next generation molecular diagnostic technology capable of simultaneously detecting multiple infectious agents from a single sample thereby improving diagnostic efficiency and clinical utility.
Individually, product groups targeting gastrointestinal infections (GI), human papillomavirus (HPV), and sexually transmitted infections (STIs) each recorded growth of more than 30%.
Improved cost structure and greater operational efficiency were cited as key factors behind the rise in operating profit. Overall profitability improved as the cost ratio for products and merchandise sales declined while selling, general and administrative expenses as well as research and development costs were managed efficiently.
By region Europe accounted for approximately 65% of total sales, maintaining its position as the company’s largest market. It was followed by Asia at 13.9%, the Americas at 13.3% and South Korea at 7.4%. Seegene continues to pursue global market expansion based on its stable Europe centered revenue base.
The company is also focusing on advancing data driven diagnostics and automation strategies. At major global conferences such as ESCMID Global 2026 Seegene introduced STAgora a real time statistical platform that connects global PCR testing data to provide analytics-based insights and CURECA a fully automated unmanned PCR testing system that automates the entire PCR testing process.
A Seegene official said “Growth centered on our non respiratory product lineup is continuing, allowing our revenue base to expand steadily.”
adding that “the company is maintaining profitability improvement through enhanced cost structure and operational efficiency.”
The official added “Seegene will continue strengthening its mid to long term growth foundation through diagnostic strategies combining data and automation.”
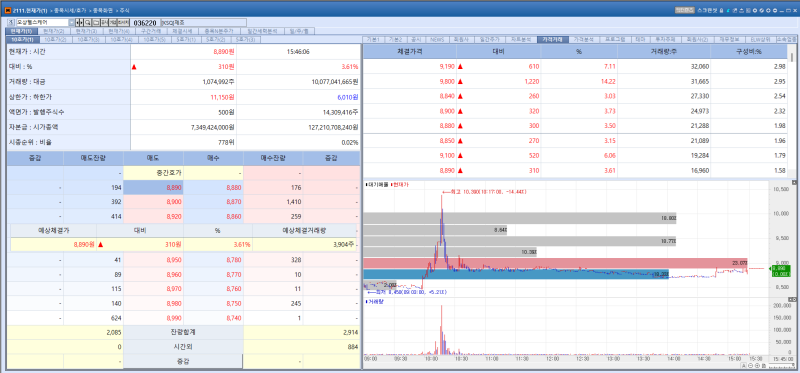
Osang Healthcare stock trend on May 11.(Image=MP Doctor)
◇Osang Healthcare Pushes Ahead With Development of Next Generation Molecular Diagnostics Platform
Osang Healthcare shares rose 3.61% from the previous trading session to close at 8,890 won on the day, extending gains for a second consecutive session. Investor sentiment was boosted by expectations surrounding the company’s efforts to develop a next generation molecular diagnostics platform.
Osang Healthcare recently announced that it had been selected for the 2026 Advanced Medical Device Research and Development Project led by the Korea Medical Device Development Fund (KMDF).
The KMDF is a joint organization established by four government ministries the Ministry of Science and ICT, the Ministry of Trade, Industry and Energy, the Ministry of Health and Welfare and the Ministry of Food and Drug Safety.
The organization supports the entire medical device development cycle including technology development, commercialization, clinical trials, and regulatory approval with the goal of strengthening global competitiveness and fostering the medical device industry ecosystem.
The selected project focuses on developing an ultra fast point of care molecular diagnostic platform for respiratory virus detection based on photothermal polymerase chain reaction (PCR) technology.
Led by Osang Healthcare in collaboration with the Korea Advanced Institute of Science and Technology (KAIST) and Samsung Medical Center the project will receive a total of 2.2 billion won in funding over three years.
Photothermal PCR is considered a next generation molecular diagnostic technology that uses light to rapidly control reaction temperatures, significantly shortening gene amplification time. Compared with conventional PCR systems the technology can greatly reduce testing time and deliver results within approximately 30 minutes.
The core technologies secured through the project are expected to be gradually applied to the company’s existing GeneFinder MX1 platform as well as to next generation molecular diagnostic systems featuring enhanced miniaturization and on site usability.
Based on these developments Osang Healthcare plans to expand into broader infectious disease diagnostics beyond respiratory infections while accelerating its global market expansion strategy.
An Osang Healthcare official said “Being selected for this project marks an important turning point in advancing our core molecular diagnostic technologies and developing next generation products.”
adding “We will move quickly to commercialize the research outcomes and strengthen our competitiveness in the global market.”
Meanwhile the company also confirmed that the diagnostic performance of its COVID-19 and influenza A/B combo test kit remains effective against the emerging COVID-19 variant Cicada (BA.3.2) which has recently been spreading domestically and overseas.
The Cicada variant has drawn attention due to concerns over vaccine efficacy, as it contains approximately 70 to 75 mutations compared with the previously dominant JN.1 lineage.
According to the Korea Disease Control and Prevention Agency (KDCA), the variant’s domestic prevalence increased rapidly from 3.3% in January to 23.1% in March.
Osang Healthcare’s COVID-19 and influenza A/B combo diagnostic kit has received authorization from the U.S. Food and Drug Administration (FDA). The product is supplied to the U.S. market through global medical device companies, while domestic sales in South Korea are handled by Yuhan Corp.










