Recent share price trend of ToolGen. A clear upward trajectory has been observed since early April. (Source: KG Zeroin MP Doctor)
◇ToolGen rises on “core patent battle” outlook; RNP scalability highlighted
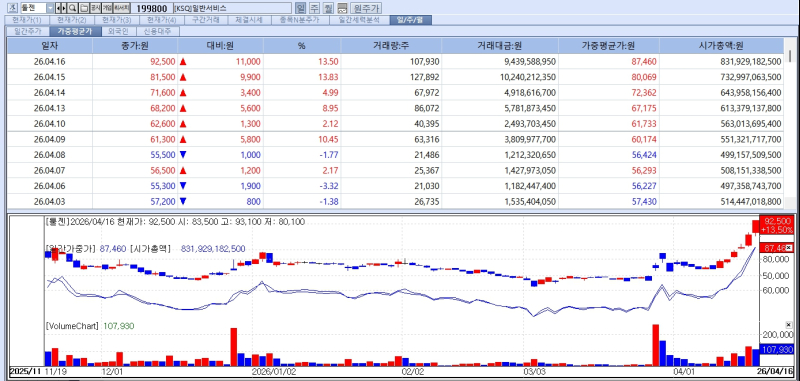
According to KG Zeroin MP Doctor, ToolGen shares rose to an intraday high of 93,100 won, marking a new 52-week peak, before closing at 92,500 won.
The company’s all-time high stands at 110,000 won, recorded on March 29, 2024, after its transfer listing from KONEX to Kosdaq in 2021. The stock later plunged to 26,650 won in August last year but has since rebounded near previous highs, maintaining an upward trend.
The turning point came in late March. On March 26 U.S. time, the Patent Trial and Appeal Board recognized the Broad Institute as the first inventor in the CRISPR-Cas9 interference proceeding involving the CVC consortium, which includes the University of California, Berkeley, the University of Vienna and Nobel laureate Emmanuelle Charpentier.
For ToolGen, the ruling effectively sets the stage for long-delayed patent disputes to move forward. Proceedings between ToolGen and the Broad Institute can resume only after the dispute between CVC and Broad enters a resolution phase.
Given ToolGen’s business model centered on patent monetization, delays in disputes have weighed on earnings visibility. The ruling has improved investor sentiment on expectations that the timeline for monetization could be brought forward.
Progress in U.S. patents related to CRISPR ribonucleoprotein technology also supported the rally. A ToolGen official said the company has 10 CRISPR RNP patent applications in the United States, with one granted in November last year and four more recently set for registration. The official added that smooth progress in U.S. patent approvals appears to be reflected in the share price.
In particular, a patent scheduled for registration on April 21, application number 19/023934, does not specify a molar ratio range. The molar ratio refers to the proportion between the Cas9 enzyme and guide RNA. By not limiting this ratio, the patent can secure broader claims covering various conditions, making design-around strategies more difficult for competitors.
The U.S. patent process generally consists of seven stages: application, publication, examination and amendment, notice of allowance, issue notification and patent grant. The four patents mentioned earlier had faced delays after the notice of allowance stage, raising market concerns. However, all four have recently received issue notifications without major issues, easing procedural risk.
◇PanGen gains on undervaluation; Samchundang hit by disclosure and trust concerns
PanGen shares rose 12.06 percent to close at 7,340 won. Investor sentiment improved after PharmEdaily reported that PanGen played a key role as a partner responsible for cell line and process development in Samchundang Pharm’s Aflibercept biosimilar program. The report highlighted PanGen’s involvement in core development stages and potential exposure to royalties and upfront payments.
The market had largely priced value around Samchundang Pharm, with PanGen’s contribution seen as underreflected. Expectations that PanGen could share in future royalty and upfront revenue streams appear to have driven a reassessment of its valuation.
In contrast, Samchundang Pharm fell 9.01 percent to 505,000 won. The stock has been on a steep decline after peaking at 1.284 million won on March 30.

◇“Controversy unresolved” as exchange review deals blow
The decline in Samchundang Pharm shares is increasingly viewed as reflecting reputational risk rather than simple profit-taking. The company attempted to address market concerns through a press briefing led by CEO Jeon In-seok on April 6 and announced on April 15 that it had initiated procedures to acquire the S-PASS oral drug delivery platform patent. However, the controversy has shown little sign of subsiding.
A key blow to investor sentiment came as the Korea Exchange classified the company’s disclosure violations as a serious matter and referred the case to the Kosdaq Market Disclosure Committee. The move signals a shift beyond simple explanations or corrections to a stage where potential sanctions are under review, significantly heightening perceived risk.
Recent controversies surrounding Samchundang Pharm have expanded from disclosure credibility issues to questions over contract structures, patent ownership and management transparency.
The company first drew criticism for disclosing a large-scale deal and earnings expectations related to an oral obesity and diabetes treatment through a press release rather than a formal filing, raising concerns over fair disclosure violations.
Questions have also emerged over undisclosed counterparties, unusual profit-sharing arrangements and relatively low initial milestone payments, casting doubt on the substance of the contract. The situation was further complicated by revelations that the S-PASS patent was originally filed by an external entity, sparking debate over ownership and technology internalization.
Additional scrutiny arose after an external figure, Seok Sang-je of Dios Pharma, took the lead in explaining key business areas during the press briefing, raising concerns over de facto management involvement. It also emerged that some patent inventors were not internal staff. These overlapping issues across disclosures, contracts, patents and organizational structure have deepened concerns over investor trust.