The benchmark KOSPI fell 2.29%, snapping a six-session winning streak, while the tech-heavy KOSDAQ dropped 2.32%. The KOSPI had climbed as high as 7,999.67 intraday a day earlier, fueling expectations for the index to break above the 8,000 point threshold before reversing sharply lower.
Against the broader decline, biotech shares outperformed. Large-cap names including LigaChem Biosciences Inc. and ALTEOGEN Inc. closed higher, while AprilBio and ToolGen both touched fresh 52week highs.
Diagnostic kit makers including Green Cross MS Corp, Sugentech, LabGenomics and Access Bio also rallied sharply.
Market participants said part of the recent profit taking in semiconductor shares appeared to rotate into biotech stocks, which had lagged behind the broader market rally.
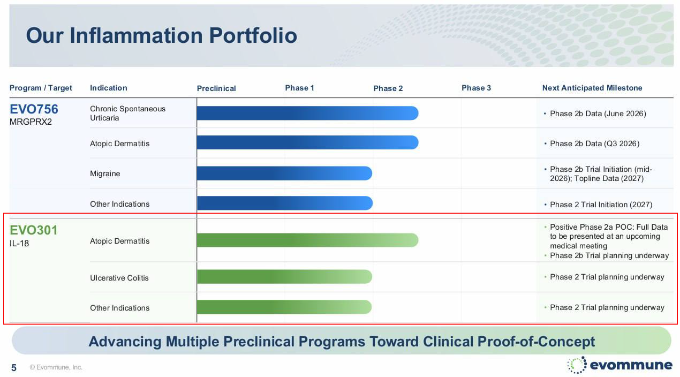
Evommune’s investor relations materials outlining development plans for inflammatory disease drug candidates. The company plans to expand indications for EVO301, licensed in from AprilBio, into inflammatory bowel disease and other indications. (Source=Evommune)
◇AprilBio Revalued as Rival Drugs Stumble
According to KG Zeroin MP Doctor, formerly MarketPoint, AprilBio rose as high as 76,700 won Tuesday marking a fresh all time high since listing.
The rally came despite news that its joint research collaboration with Yuhan Corporation had ended recently. Investors instead focused on the value of its existing license out pipeline. A key driver has been a series of setbacks among competing drug candidates.
APB-R3, an atopic dermatitis treatment licensed out by AprilBio to U.S. biotech company Evommune under the name EVO301, has gained renewed attention following disappointing clinical results from next generation rivals.
Sanofi’s dual target drug amlitelimab positioned as a potential successor to blockbuster Dupixent, recently failed to meet the primary endpoint in a Phase 2b trial for atopic dermatitis, shifting market sentiment.
Dupixent generates annual sales of roughly $17.8 billion globally, but demand for next-generation therapies has persisted due to incomplete remission rates and treatment-resistant patients.
EVO301 drew attention earlier this year after achieving statistical significance in a Phase 2a study. Unlike existing IL-4 and IL-13-focused therapies, the drug directly targets IL-18, a differentiated mechanism that some analysts believe could expand treatment options for patients unresponsive to current therapies.
Evommune also recently disclosed plans to expand EVO301 into additional indications including ulcerative colitis, cardiovascular inflammatory diseases and food allergies, according to company investor relations materials.
Sentiment has also improved in thyroid eye disease or TED after several FcRn targeting therapies, including HanAll Biopharma’s batoclimab and Argenx’s efgartigimod reported disappointing clinical outcomes in the indication.
AprilBio’s APB-A1 approaches TED through a CD40L mechanism rather than FcRn inhibition. The candidate is currently undergoing a Phase 1b study through Lundbeck, where signals of reduced eye protrusion have reportedly been observed in some patients.
Lundbeck has already decided to advance the program into Phase 2 before the release of final data.
Investors are closely watching detailed results expected to be presented at ENDO 2026 in June, which many believe could serve as a key catalyst for a revaluation of AprilBio.
◇ToolGen Surges Fivefold in Nine Months
ToolGen also hit a new 52-week high Tuesday, climbing to 142,000 won intraday. The stock has surged more than fivefold in just nine months after falling to the 20,000 won range in August last year. Prior to this year’s rally its post transfer listing high on the KOSDAQ stood at around 110,000 won recorded in March 2024.
The rally has been fueled largely by progress in the U.S. CRISPR patent dispute. The U.S. Patent Trial and Appeal Board in March recognized the Broad Institute’s priority claim, accelerating long delayed interference proceedings related to CRISPR-Cas9 patents.
Investor expectations for patent monetization also increased after ToolGen recently secured three U.S. patents related to CRISPR ribonucleoprotein or RNP technology.
The company is now viewed as diversifying its revenue base beyond foundational eukaryotic cell patents into RNP related intellectual property. ToolGen Chief Executive Yoo Jong-sang previously said during an investor relations event that the company had evolved from relying on a single foundational patent platform to securing a second growth pillar through RNP patents.
He added that the expanded portfolio could attract not only drug developers but also xenotransplantation firms and agricultural seed research companies. Expectations surrounding the company’s seed business also contributed to the rally.
ToolGen recently said it is negotiating license out agreements with global seed companies centered on drought resistant pepper varieties. The company said discussions accelerated after restructuring its seed pipeline to focus more on profitability.
Drought and herbicide resistant crops are increasingly seen as high demand areas amid climate change and food security concerns. ToolGen has also expanded into oil crop development for sustainable aviation fuel applications.
◇Hantavirus Concerns Lift Diagnostic Kit Stocks
Concerns over a hantavirus cluster outbreak also fueled gains in diagnostic kit related stocks. Additional infections were recently reported among passengers who disembarked from the cruise ship MV Hondius boosting speculative interest in related stocks.
Access Bio surged 29.89%, nearing the daily price limit while Green Cross MS rose 18.28%, Sugentech gained 13.22% and LabGenomics advanced 7.77%. However, no South Korean company has officially announced plans to develop hantavirus diagnostic kits or vaccines.
A Green Cross MS official said the company currently has no plans to develop related diagnostic products. The World Health Organization has also assessed the likelihood of a COVID-19-scale pandemic as low.
While the Andes virus strain circulating in South America has shown rare cases of human-to-human transmission, experts say its transmissibility remains limited. Hantavirus is primarily transmitted to humans through rodents and can rapidly progress from fever and muscle pain to acute respiratory distress syndrome or multi-organ failure within hours.
No effective antiviral treatment currently exists, making early diagnosis critical. Moderna recently disclosed that it is in the early stages of hantavirus vaccine development but no domestic companies have formally announced related vaccine or diagnostic projects.
Industry officials said the recent rally in diagnostic kit stocks appeared driven more by short-term thematic trading than by fundamental earnings expectations. An executive at a domestic diagnostic kit developer said hantavirus is not considered technically difficult for diagnostic kit development and that products could likely be developed relatively quickly if the government were to encourage such efforts from a national security perspective.